Similarly, IMpower 110 demonstrated atezolizumab alone did not differ in OS compared to chemotherapy in the PD-L1 expression ≥1% group, but significantly prolonged OS in the PD-L1 expression ≥50% group ( 5). In addition, overall survival (OS) in the ICI group was significantly longer than in the chemotherapy group in all three populations with different PD-L1 expression, and the difference is largest in the PD-L1 expression ≥50% group ( 4). In KEYNOTE 042, compared with chemotherapy, progression-free survival (PFS) of pembrolizumab monotherapy was not statistically different in the PD-L1 expression ≥20% and≥1% groups, but it was significantly longer in the PD-L1 expression ≥50% group. The KEYNOTE-024 study showed that, as a first-line treatment, pembrolizumab monotherapy remarkably improved the treatment efficacy in patients with advanced NSCLC and PD-L1 expression ≥50% compared with chemotherapy ( 3). For patients with NSCLC, immune checkpoint inhibitors (ICIs), including programmed death-1 (PD-1), programmed death-ligand 1 (PD-L1), and cytotoxic-lymphocyte antigen 4 (CTL-4) inhibitors, have brought great progress. A subset of NSCLC patients with driver mutations is able to benefit from targeted therapy ( 2) however, platinum-based chemotherapy is still received by patients who lack driver mutations. Non-small cell lung cancer (NSCLC) accounts for 85% of all primary lung cancers ( 1). Guangzhou Institute of Respiratory Health, State Key Laboratory of Respiratory Disease, First Affiliated Hospital, Guangzhou Medical University, 151 Yanjiang Road, Guangzhou 510120, China. 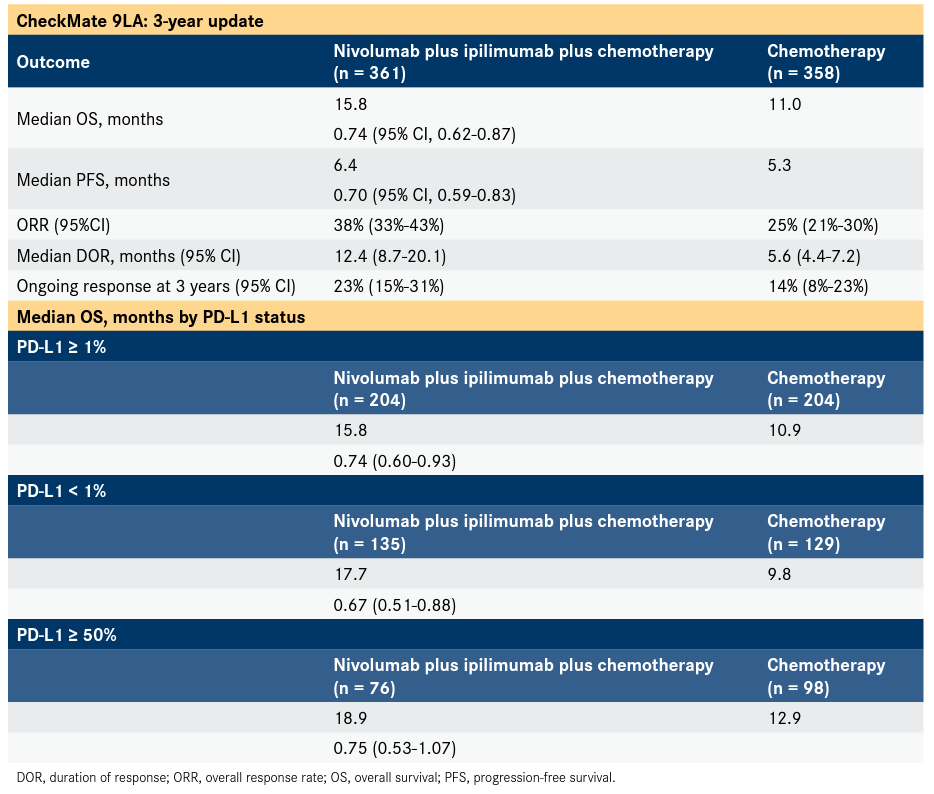 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
